Home
Study Details
-
Objective
• The objective of the study is to compare angiographic outcomes of Magic Touch sirolimus coated balloon (Concept Medical) versus SeQuent Please Neo paclitaxel coated balloon (B.braun) for the treatment of de novo coronary artery lesions in small vessels (≤2.75 mm) with respect to Net Gain (mm) at 6 months follow-up
• A prospective, randomized, multicenter study in subjects with small vessels, i.e. at least one de-novo lesion in a small vessel (≤2.75mm)
•OCT will be performed post pre-dilatation (guidance) prior to DCB treatment
• The DCB balloon size will be selected based on OCT measurements
-
Endpoints
Primary Endpoints:
The primary endpoint is in-segment (balloon treated area) Net Gain (mm) at 6 months post-procedure.Secondary Endpoints:
Device success (Lesion based); Procedure success; Angiographic outcomes (late lumen loss, minimal lumen diameter, percent diameter stenosis, restenosis rate); Device oriented Composite Endpoint (DoCE / TLF) defined as the composite of cardiac death, TV-MI, and clinically indicated target lesion revascularization (TLR); Acute/subacute/early/late vessel closure/thrombosis. -
Clinicaltrials.gov
NCT03913832
Study Countries

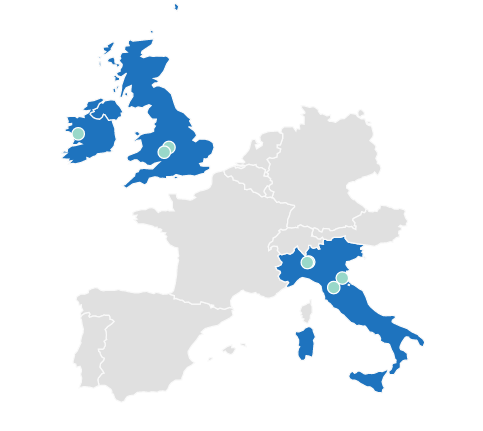
Patients Enrolled
121
Italy
-
Study PI
– Prof. Antonio Colombo
-
Maria Cecilia Hospital, Cotignola
– Dr. Roberto Nerla
-
AOU Careggi, Florence
– Prof. Carlo Di Mario
-
Policlinico San Donato, San Donato
– Prof. Luca Testa
-
Humanitas Research Hospital IRCSS, Rozzano
-Dr. Bernhard Reimers
UK
-
Heartlands Hospital, Bordesley Green Birmingham
– Dr. Sandeep Basavarajaiah
Ireland
-
NUI Galway, Ireland
– Dr. Faisal Sharif
Sponsor
Concept Medical B.V
Hogebrinkerweg 33,
3871KM Hoevelaken,
The Netherlands.
Concept Medical Research Pvt. Ltd.
Office 901~903, Trinnity Orion,
Beside Jolly Residency, Vesu,
Surat-395007, Gujarat, India.
Concept Medical Inc.
5600 Mariner ST, STE 200,
Tampa, FL 33609,
USA
Concept Medical PTE Ltd
3 Shenton Way, #12-08 Shenton House,
Singapore 068805







